Dr Michelle Tempest and George Holden of Candesic, investigate the emerging market and white space in digital clinical trials and discuss how mixing old and new pathways will disrupt the sector and create global investment opportunities
Innovation is for the curious and those who love to experiment. Clinical trials are all about bringing innovation from bench to bedside. Historically, they have been restricted to the confines of physical buildings, headquartered in academic centres and heavily paper based. The result has been several unintended consequences, such as lack of diversity among human sample sets, high costs, and very long timelines. Some 80% of clinical trials fail to meet initial recruitment targets and deadlines1.
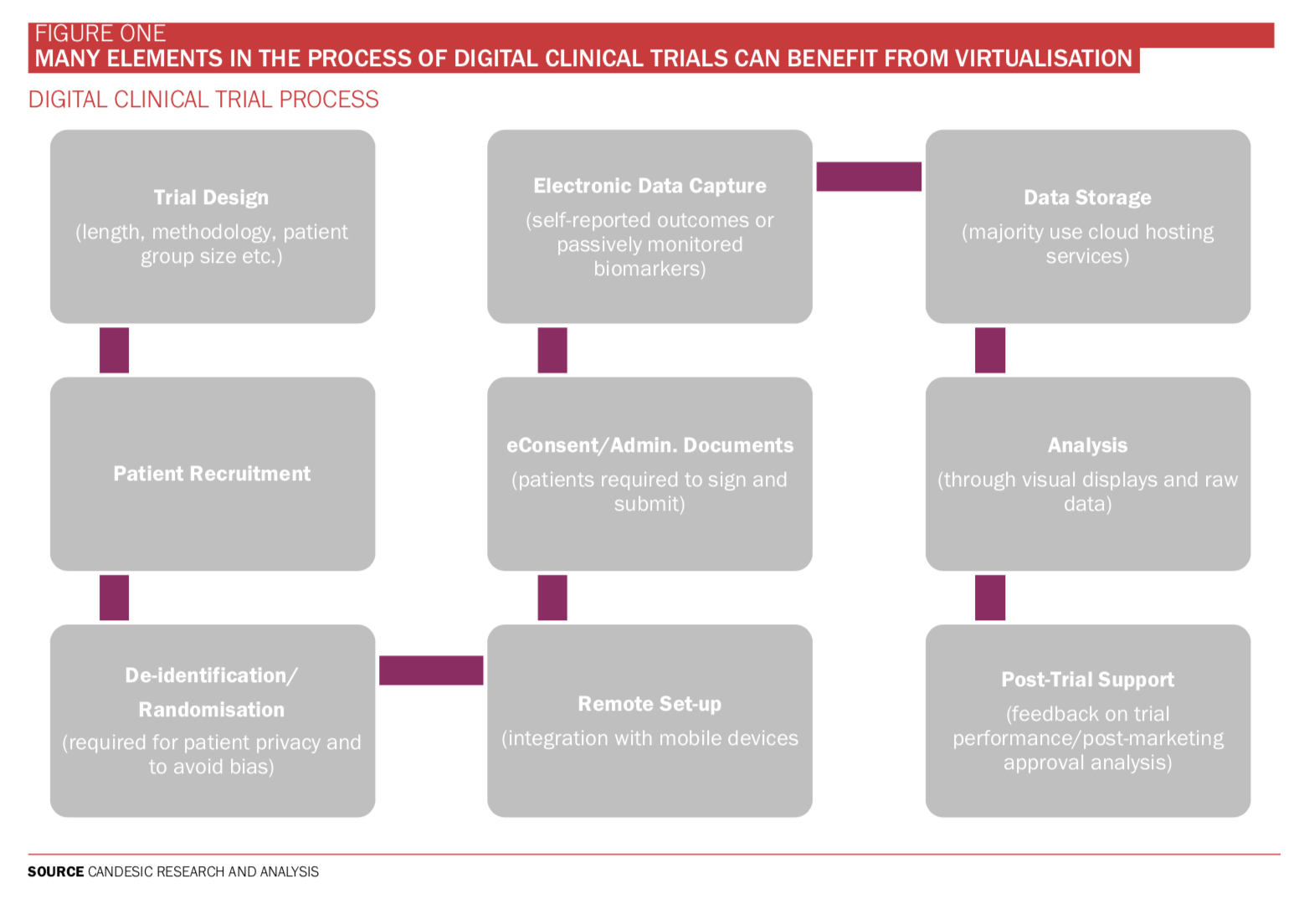
This is not good for science nor advancement. It also reduces access to the so-called ‘Trial Effect’ – where outcomes can be better for all enrolled. Now is the time to rocket boost digital democratisation of clinical trials and accept that not everything in trials can be digitalised. Clever boffins remain vital, but ideas will spread faster by leveraging digital recruitment, bespoke electronic trial IT, home monitoring, data storage and AI big data analysis (see Figure One).
The clinical result will reduce bias, get faster results, and make the entire value chain more cost effective – hence a win- win for all those thinking of investing in this sector.
Rocket boost digital enablement
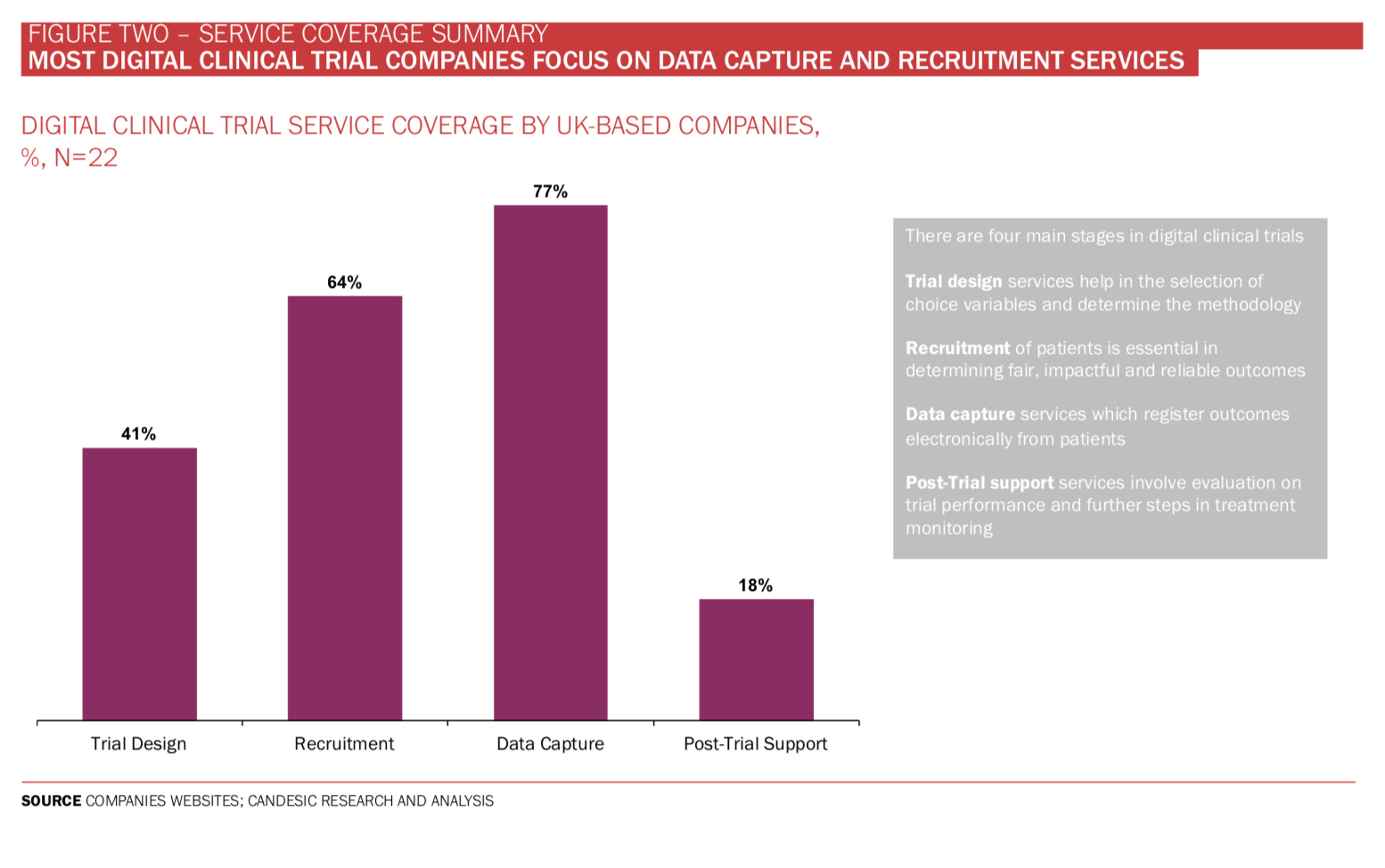
Digitalising services which support clinical trials can unlock a host of benefits for patients and clinicians. From our analysis of companies supporting decentralised digital trial platforms (see Figure Two), many find suitable matches between patients and trials – which can be done globally. An additional service is assisting in patient randomisation, helping to remove bias from trials. Then there are other advantages of digital support services and a whole host of cybersecurity, digital twin analysis and AI which are beyond the scope of this article.
Here in the UK, alongside some of the larger fully digital and hybrid clinical trial technology, there has there been a surge in start-ups – a silver lining of Covid-19. The pandemic hit the world of clinical research hard and necessitated a rapid pivot towards decentralised, remote trials.
One such company which has reaped the benefits of being ahead of the curve is Patients Knows Best (PKB). Founder and CEO Dr Mohammed Ubaydli told us: ‘PKB now has international reach and is integrated into the NHS app meaning we can help offer every person with an NHS number the opportunity to be part trials if they wish to be, in an easy to access way.’
While patient recruitment is arguably the most impactful factor, the entire process from trial design through to post-trial support can be made more productive and cost-effective through digital support services. For example, electronic data capture software is a broad term and can provide a simplified record of patient out- comes, both those which are self-reported and passively monitored, for instance, through integrated devices or wearables. This cuts out the two-stage traditional process of manual notetaking followed
by electronic transcription. Passive biomarkers can be constantly monitored for a large sample of patients, providing increasingly accurate monitoring data during trials, uninfluenced by patient biases inherent in self-reporting. Furthermore, they could help improve patient retention by reducing the burden of self-reporting. Estimates show patient dropout increased from 15% in 2012 to approximately 20% in 20192.
Many companies already offer integration with mobile devices but there is still space for this to grow. The wearables market is expanding rapidly (13% CAGR until 2024)3, and they are likely to be increas- ingly used after securing clinical grade regulatory approval. When we spoke to Dan Vahdat, CEO and founder of Huma, he said: ‘We are increasingly contracting with pharma to deliver real time home monitor- ing for their clinical trials. It’s something we have been developing for many years and as a result we are a trusted partner.’
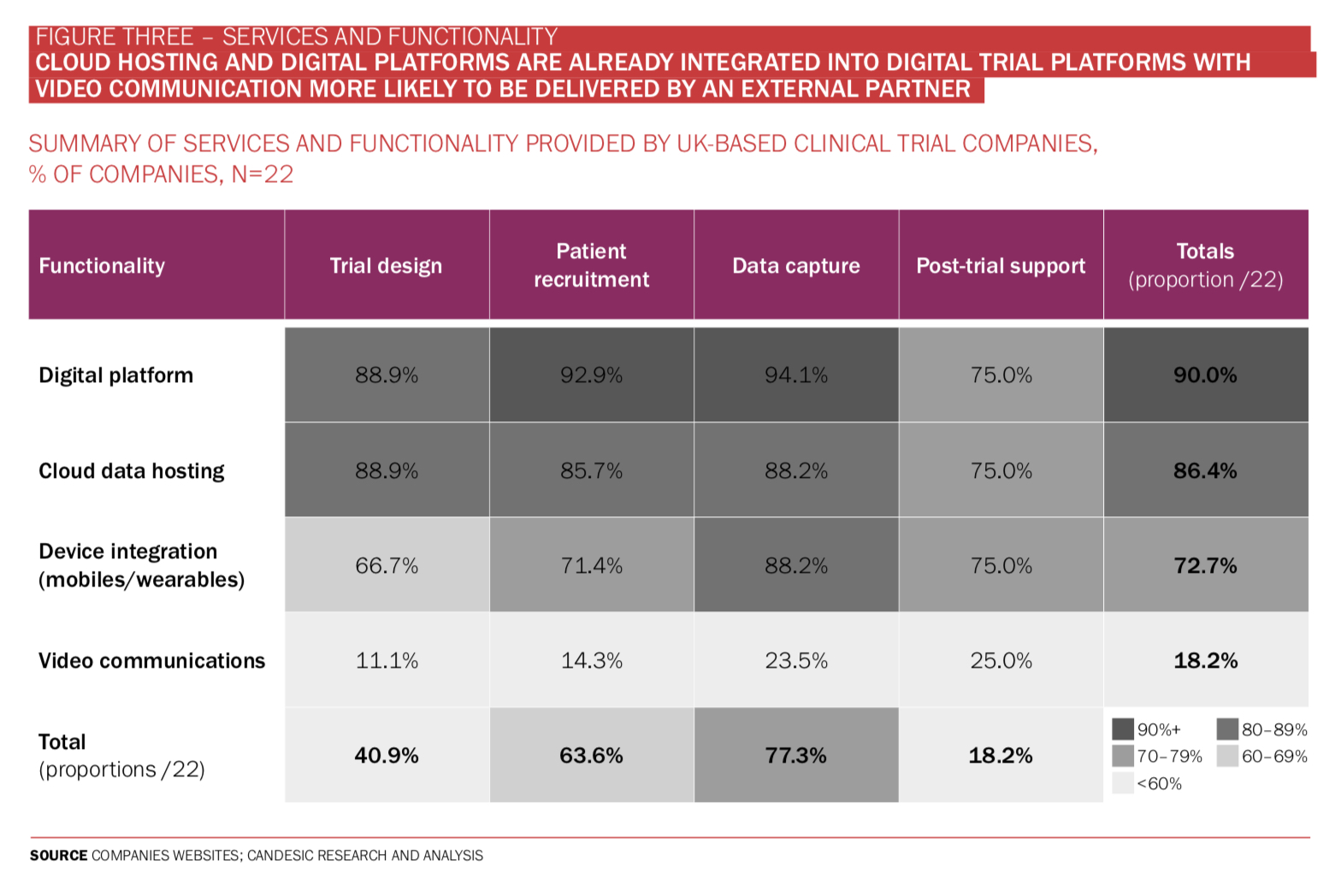
Among the UK-based digital clinical trial support companies, electronic data capture is offered by 77% (see Figure Three) alongside a digital platform, data analysis and visualisation of data. Dr Elin Haf Davies, founder and CEO of Aparito, says: ‘Pharmaceutical companies and reg- ulators are relatively risk averse to protect the safety of patients while taking part in clinical trials. In parallel, however, the need for patient centric clinical trials is becoming an ethical imperative. That means enabling at home assessment of drug and disease impact on patients via standardised cap- ture of clinical outcome assessments (COA) and patient reported outcomes (PRO).
This includes digital biomarkers via video and voice assessments. Our software platform Atom5TM has been developed and validated to regulatory standards for such an approach, making the patient interface and usability key to engagement.’
To date, few companies support post-trial services such as evaluative feedback on trial performance and next steps. Perhaps the lack of coverage is indicative of a lack of historical digital trial information with which to base a comparison. In which case, expect to see this service provision grow significantly as more knowledge on what constitutes a successful digital trial accumulates.
Differentiation and unique selling points
It comes as no surprise that broad functionality of software was key for customers. Alongside this they mentioned transparency and interoperability.
Of the companies analysed, 86% offered integrated cloud hosting services to aid safe and scalable storage of gathered data (see Figure Four). For video consultation or communication, only 18% of companies analysed offer this functionality. This is likely due to secure applications for video communications being freely available, albeit not within the same interface that are used and not always with the necessary privacy compliance. It is also possible that the market for telehealth is itself still growing and insufficient time has elapsed for us to see interoperability between the markets.
What the future holds
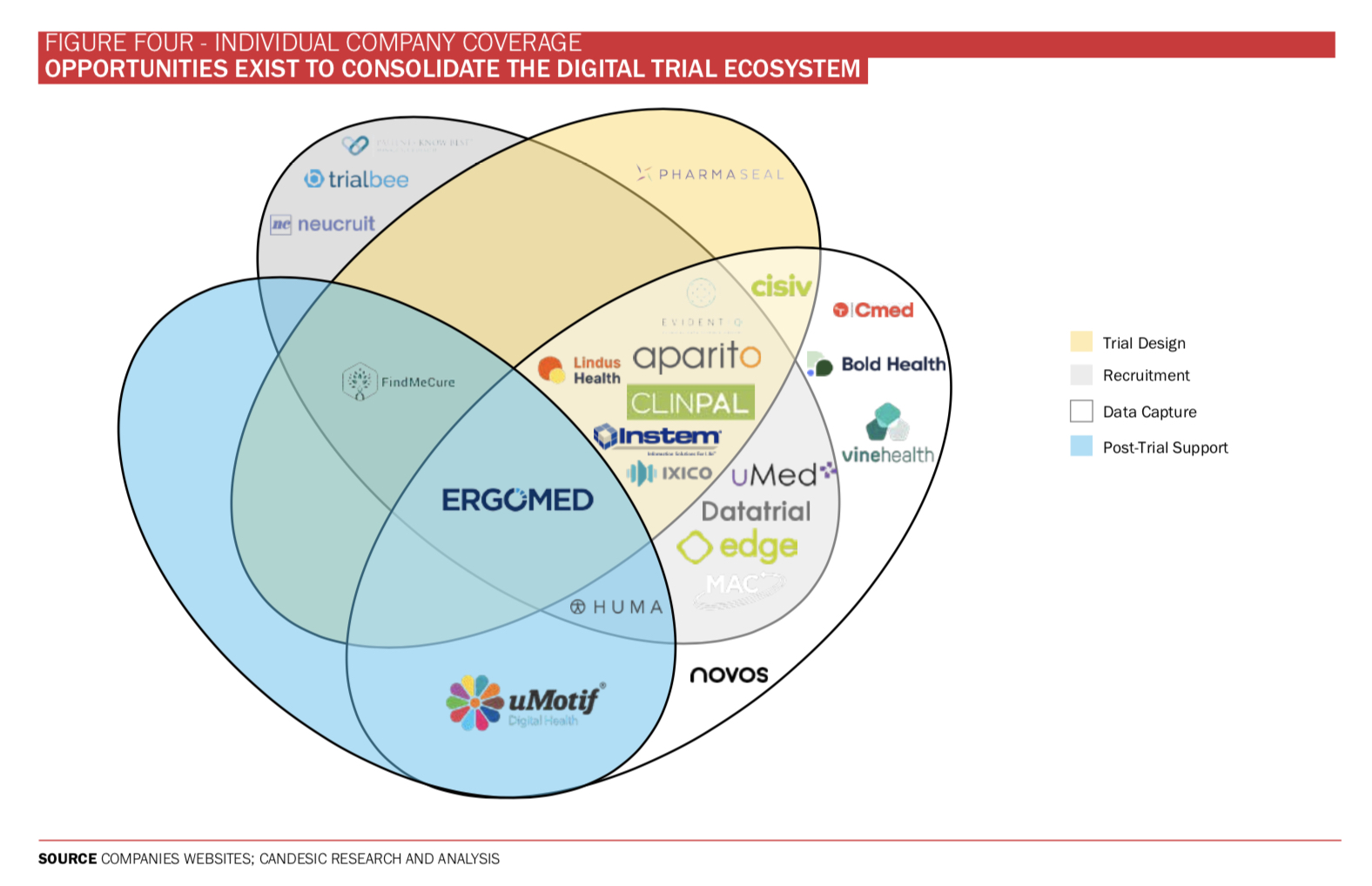
Looking ahead, there is likely to be a dichotomy between large Clinical Research Organisations (CROs) able to fully fund and pivot their services to decentralised processes alongside smaller disruptors looking to offer value software packages. Last year saw record levels of investment in the space, including Medable, the US-based decentralised clinical trials platform, which raised over $380m.
There are opportunities to consolidate this market, as well as appetite for CROs to acquire successful disruptors as they mature.
This thesis is already converting into reality with plenty of M&A activity in recent years such as Signant’s acquisition of Vir-Trial in 2020 and ICON’s acquisition of PRA Health Sciences in 2021. Ben Maddison, a director within Stifel’s global healthtech team notes: ‘R&D within healthcare is striving to become more efficient and the digital enablement of clinical trials is a core part of achieving this. In a rapidly growing and maturing market, where investment remains at record levels, we are witnessing heightened strategic and private equity interest in acquisitions. We see this trend continuing.’
In summary, the Candesic review of this market flags the excitement in this sector which sits at the very heart of life sciences and clinical care. Influxes in investment into digital services are no longer just desired but urgently required to expedite novel treatments and improve outcomes at scale and pace globally.
NOTES 1 Brøgger-Mikkelsen M, Ali Z, Zibert JR, Anders- en AD, Thomsen SF. Online Patient Recruitment in Clinical Trials 2020 2 Couch Health 3 Cision PRNewswire








 ©2024 All rights reserved LaingBuisson
©2024 All rights reserved LaingBuisson 


